Latest Licensure Breaks Records for Ho-Hum Reception, yet What Exactly Is There to Be Excited About?
A Co-Formulation Elation?
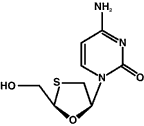
3TC
|
Emtriva
|
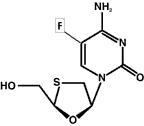
Earlier this month, the fluoronated 3TC agent, FTC (emtricitabine, Emtriva) received FDA approval for the treatment of HIV in adults. The latest once-daily nuke on the block (now that Gilead has acquired the drug from Triangle Pharmaceuticals), lends itself handsomely to a fixed dose co-formulation with Gilead’s other QD offering: tenofovir (Viread); FTC is also in Phase III clinical studies for the treatment of chronic hepatitis B. Excerpts of TAG’s position paper on FTC, prepared by Antiviral Project Director, Rob Camp, appear below.
FTC is a once-daily (QD) thiacytidine nucleoside analogue developed by Triangle Pharmaceuticals. The compound, 2′-3′-dideoxy-5-fluoro-3′-thiacytidine, was licensed from Emory University in 1996. It comes in 200 mg capsules and should be stored at room temperature. There are no food restrictions with FTC administration. FTC has been developed quite slowly. What may have been the most interesting thing about this drug (i.e., once daily dosing) is less interesting now that 3TC is licensed as such.
People with the M184I/V mutation associated with 3TC (Epivir) will not benefit from FTC, given that the M184 mutation confers high-level resistance to FTC. For this reason, it is important that persons with detectable viral loads who plan to switch from 3TC to FTC have genotypic testing performed to determine whether the M184V mutation is present. (Of course, a patient’s treatment history is also extremely important.) If 3TC has failed in the past, the 184 mutation is archived, thus rendering FTC ineffective.
How Effective Is FTC for the Indication in Question?
In all studies to date, including the FTC-302 study (which was stopped due to liver issues, later shown not due to FTC), this drug has been seen as a competent first-line cytidine analogue, the main difference being that it is dosed once a day. Interestingly, Triangle/Gilead conducted head-to-head comparison studies with other nucleoside analogues. Unlike abacavir where the development philosophy was to demonstrate its superiority to a combination of two nucleoside analogues (AZT and 3TC), the FTC development strategy involved comparing it directly with d4T and 3TC, utilizing a “non-inferiority” design.
What Are the Real Side Effects?
Again, FTC deserves a thumbs up. Although new and more horrific side effects are discovered over time with most drugs, because FTC is so similar to 3TC, the side effect profile is probably well-characterized. Although neither drug was ever studied in monotherapy for extended lengths of time, 3TC has shown over time that headache, malaise/fatigue, nausea, sleep interference and dizziness are the main side effects, all hovering around 5%. Lab abnormalities do not reach 2% at 24 weeks. FTC side effects should be very similar.
Are Things Not Being Said?
Triangle could have probably been a bit more imaginative in their trial designs, but there seem to be only a handful of questions unaddressed:
- One issue not looked at yet is FTC’s effect in pregnancy.
- Pediatrics trials done are being repackaged and resubmitted at a later date. When and why?
- There also was a recent charge from the FDA that the Abbott production facility where FTC was being produced was not maintaining standards of quality. The Abbott facility is no longer producing FTC.
- The dose-ranging study is only in press now. Why was it submitted so late?
- Liver toxicity may take longer to see than the observation period of these registration studies.
- Trials should have been stratified for hepatitis B serostatus.
Efficacy Studies
In FTC-301, antiretroviral-naive HIV-infected people with viral loads >5,000 copies/mL were randomized to receive FTC once daily or d4T twice daily and matching placebo, as well as open-label Videx EC (ddI) and Sustiva (efavirenz). Study endpoints included measurement of viral failure, the frequency of genotypic mutations at time of failure and the CD4 change from baseline between the two groups. The primary measure of analysis for this study was the percentage of patients with plasma HIV-1 RNA <50 copies/mL at Week 48. The study was powered to be able to demonstrate equivalence between FTC and d4T within a difference of 12.5% as well as to detect differences between treatment groups of 15%.
Five hundred and seventy-one people were randomized and received at least one dose of randomized study medication. At baseline, the median viral load was 4.9 log copies/mL and the median CD4 count was 288 cells/mm3. The proportion of people having viral failure through Week 48 was 5.3% in the FTC group and 12.7% in the d4T group (p<0.01). The mean increase from baseline to Week 48 in CD4 was significantly greater in the FTC group (+153 cells/mm3) than the d4T group (+120 cells) (p<0.05).
Also measured was efficacy failure — defined as virologic failure, death, progression to CDC class C event, or loss to follow-up — which occurred in 18% of people in the d4T group and 9% of people in the FTC group through Week 48 (p<0.01). In terms of the proportion of people with undetectable viral loads at Week 48 using an intent-to-treat analysis (non-completer equals failure): 80% in the FTC group and 67% in the d4T group had viral loads below 400 copies/mL (p<0.001), and for plasma viral load <50 copies/mL, 74% FTC, compared to 58% d4T, got there after 48 weeks of treatment (p<0.0001).
Genotypic analysis was performed on all of the 49 individuals with confirmed virologic failure through Week 48 (35 d4T, 14 FTC). Of the 35 genotypic evaluable volunteers failing d4T, 34 (97%) had mutations in the HIV polymerase gene as compared to 71% (10/14) from the FTC subgroup (p=0.019). The M184V mutation was observed only in the FTC subset, 43% (6/14), while the thymidine analogue mutations (“TAMs”) were observed in 7% (1/14) of the FTC group and 20% (7/35) of the d4T group. Although these results show that FTC was statistically superior to d4T, it is worth reminding that the combination ddI + d4T is not regularly recommended.
In another “non-inferiority” study, the French ANRS 099 or ALIZE, people receiving a protease inhibitor-based regimen with plasma HIV-RNA level <400 copies/mL were randomized to continue their regimen (C) or to switch to once-daily combination (5 pills per day) of FTC, ddI, and efavirenz (once daily). Intent-to-treat on available data, on treatment on available data and intent-to-treat with (missing=failure) analyses were conducted.
A total of 355 people were randomized; 86% were male with a median age of 41, a median duration of PI use of 35 months, and a median CD4 count of 540 cells/mm3. Of course, today, at 540 CD4s, these people would probably be offered a treatment interruption of some sort instead of rolled into another new treatment protocol of 48 weeks. The proportion (98%) of people with virologic success at Week 48 was 92% for the “C” group (continue) and 94% for the FTC group (ITT).
There was a statistically significant difference in the proportion of people having plasma HIV-1 RNA <50 at Week 48 (95% on once daily vs. 87% on C, p=0.01). Median CD4 count increase was similar between groups. Rates of treatment discontinuations were “low” (12.4% and 10.1%, no statistical difference). A significant increase in median fasting HDL cholesterol levels was observed in the QD group as compared to the C group. Other metabolic parameters remained similar between groups throughout the 48 weeks of the study. People who simplified to this once-daily regimen had an improved outcome compared to subjects remaining on more the complex protease inhibitor-based regimens.
FTC-303 was a randomized, 48-week, open label equivalence trial in which people with HIV-1 RNA <400 copies/mL either continued their 3TC regimen or switched 3TC 150 mg twice daily to FTC 200 mg once daily while continuing the other medications in their regimen. People with plasma HIV-1 RNA <400 copies/mL at Week 48 were offered FTC as part of their HAART regimen in protocol FTC-350.
Out of 294 people originally randomized to FTC in study 303, 227 (77%) had HIV-1 RNA <400 copies/mL at Week 48. Of these, 215 continued in rollover study FTC-350; 152 of 294 (51%) maintained suppression of HIV-1 RNA <400 copies/mL and 139 (47%) <50 copies/mL through Week 120 (2.3 years). Through a median follow-up of four years, the probability of virologic failure (>400 copies/mL) was 11% while prescribed FTC.
FTC was well tolerated throughout the study. The probability of treatment limiting adverse events was <13% at year 4 of follow-up. The annualized rate of the most common treatment-emergent Grade 3 or Grade 4 laboratory abnormalities was 7% for creatine kinase and 5% for hypertriglyceridemia, with the annualized rate of all other laboratory abnormalities <2% after 4 years of follow-up. Asymptomatic and transient elevations in CPK accounted for more than 2/3 of the overall Grade 4 adverse events. Everyone continued on the background regimen that was part of the stable 3TC regimen at the start of FTC-303. This regimen included AZT or d4T as well as an NNRTI or PI.
Studies to Be Done
- A pharmacokinetic assessment in people with different stages of hepatic impairment.
- Are there plans to do AZT+FTC studies due to neurological issues seen with AZT+3TC?
- Is there a clinical benefit to keeping the 184V mutation active?
- In a treatment interruption setting, should FTC be stopped before the other drugs, not unlike 3TC and the NNRTIs?
- ACTG studies 5015 (accelerated disease and aging) and 5073 (a DOT study) are fully enrolled and should be reporting results soon.
Expanded Access
There has been no expanded access for FTC. We would like to express our regret at the sponsor’s unwillingness to test this drug in real life prior to approval. Even an open-label pseudo-expanded access scheme (where the issue is not the life and death of patients) would have been helpful in better defining some of the outstanding questions as enumerated in this report.
The author would like to thank Francois Houyez for his collaboration on this paper.