Bold strategies are required to remedy frequent stock-outs and supply interruptions
By Lindsay McKenna
Drug shortages, especially of tuberculosis (TB) drugs, have become increasingly common in the United States. Over the past year alone, the U.S. Centers for Disease Control and Prevention (CDC) has reported shortages (also referred to as stock-outs or supply interruptions) of various TB products including second-line injectables (capreomycin and amikacin), required to fight drug-resistant TB (DR-TB), and tubersol and aplisol, important products for TB diagnosis.
Shortages of isoniazid, one of the most powerful drugs to fight both drug-sensitive TB (DS-TB) and latent TB infection (LTBI), have led to rationing in some regions. “That put me in a really uncomfortable position as a providing physician, but also as the health official telling doctors they had to pick and choose who gets isoniazid and who doesn’t,” said Charity Thoman, MD, MPH, deputy director of the Santa Barbara County Department of Health, which began rationing its supply of isoniazid in January. “For me, that created an ethical dilemma, because that’s not what you sign up to do as a doctor. We should be providing treatment for all our patients.“
Domestic TB drug shortages are a recurring issue, and existing stopgap measures are inadequate, threatening the United States’ status as a model for TB elimination. Over the last decade, U.S. TB drug shortages have tripled. In addition to the aforementioned drugs, the CDC has reported interrupted supplies of injectable rifampin, fixed-dose combinations of isoniazid and rifampin (used to treat and reduce the pill burden for DS-TB and LTBI patients), rifabutin (commonly used to treat TB in HIV-coinfected patients), ethambutol (one of four drugs that make up first-line therapy), and ethionamide, streptomycin, and cycloserine, which are used to treat DR-TB.
To add insult to injury, it is not unprecedented for manufacturers to take advantage of ongoing shortages to increase prices. In 2007, when Akorn took over manufacturing of capreomycin, the price for a one-gram vial increased thirtyfold from US$11.71 to US$300. During the isoniazid shortage earlier this year, pharmacies reported that the price of a 30-pill batch of 100 mg tablets would increase from US$35.51 to US$1,309.94—a 3,589 percent price increase. Fortunately, after much vocal protest and the promise of public shaming, isoniazid manufacturers did not follow through. But even small price increases from pre-established levels can devastate state programs working within cash-strapped budgets.
Though President Obama issued an executive order in 2011 directing the U.S. Food and Drug Administration (FDA) to notify the Justice Department of suspicious pricing, the FDA states that pricing issues are outside of its purview. Nonetheless, the FDA needs to take accountability for and address drug pricing, and work with the Justice Department to reprimand all manufacturers who increase prices during ongoing and future drug shortages. The FDA’s dismissal of responsibility to address drug pricing highlights a larger issue: the FDA and the CDC continuously pass the buck when it comes to implementing long-term solutions to recurring drug shortages. While the FDA ultimately acts as gatekeeper for manufacturers looking to procure drugs to programs in the United States, the CDC has the capability to establish a national drug procurement system. Yet the number of FDA-approved TB drug manufacturers continues to decrease, and fragmented, state-by-state procurement continues. The muddled division of responsibility allows each to off-load accountability to the other, ultimately leaving programs and patients to pay the price.
The effects of drug shortages are burdensome to TB programs and patients: they consume program staff time, cause patients to miss doses (which can lead to the development of drug-resistance), force patients to switch to inferior regimens, and require the use of more expensive drugs. Drug shortages are also problematic for research programs. If study drugs are not purchased up front for use in clinical trials, shortages can result in research interruptions and delays, which have the potential to threaten the validity of study results. The negative impact of shortages is compounded by a lack of effective communication to TB program managers, researchers, and care providers.
Although shortages are reportable to the FDA under Title X of the 2012 FDA Safety and Innovation Act (FDASIA), this legislation is weak, and passive at best. If a manufacturer fails to report an impending drug shortage, its punishment is an FDA-issued noncompliance letter, which is made available to the public. The FDA does not work closely with the CDC to communicate shortages systematically and effectively to those who are actually responsible for treating patients, or to gather information from providers and pharmacists to determine if there are local supply issues.
One way to improve close collaboration between the CDC, the FDA, providers, and pharmacists, and to prevent programs and patients from suffering the effects of shortages, is for the CDC to establish a central emergency stockpile of TB drugs at the U.S. Department of Health and Human Services Supply Center. Programs can then draw from the emergency stockpile during shortages. Additionally, government resources and capacity for preventing and monitoring shortages must be increased, and interagency coordination and communication must be improved. Manufacturers should inform the FDA of impending shortages or plans to withdraw products much earlier, and the FDA should vigorously enforce Title X of the FDASIA.
According to manufacturers, there are several causes of drug shortages [see figure 1]. But the underlying issue is the small number of manufacturers producing FDA-approved TB drugs and active pharmaceutical ingredients (APIs). Capreomycin and amikacin each has a sole manufacturer with FDA approval. This leaves programs reliant on one manufacturer, which can exit the TB space at any time without repercussion. This lack of manufacturing diversity is partially due to the fact that most TB drugs are older and off-patent, so the TB market—especially the small number of cases of DR-TB—is not viewed to be profitable for manufacturers, and few of them are willing to take on the expense and challenge of seeking FDA approval.
Figure 1. A Recent History of TB Drug Shortages
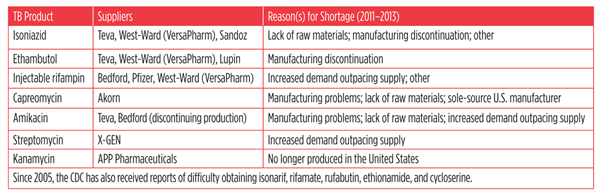
NOTE: A majority of manufacturers obtain active pharmaceutical ingredients (APIs) necessary to make drug from other manufacturers. When there is a problem with API production, the drug manufacturers report a “lack of raw material” as the reason for supply interruption or shortage; this becomes a serious threat to TB patients and the public when only one FDA-approved API manufacturer exists (as is the case for capreomycin). “Manufacturing problems” are reported when the production line for a specific drug breaks down, halting manufacturing. The effects of manufacturing problems on programs are delayed until after existing program drug stocks run out. “Increased demand outpacing supply” is often seen when one manufacturer experiences a production-line breakdown, leaving the other manufacturer(s) to cover the gap left in program supplies. Similarly, if there are already few manufacturers and one discontinues production of a drug (“discontinuation of manufacturing”), a hole is created that the other manufacturer(s) may struggle to fill.
To make matters worse, starting in October 2013, the FDA will increase application fees for manufacturers seeking approval to make generic drugs, potentially further discouraging manufacturers from entering this already neglected space. The FDA should exempt TB drugs from these fee increases and provide incentives (such as tax credits) to diversify TB product manufacturers. Making TB an attractive market is of particular importance, as the FDA has no authority to mandate manufacturers to make drugs or to continue to produce older products, even if they are medically necessary and relevant to public health.
Alternatively, if the FDA were to accept approval from other Stringent Regulatory Authorities (SRAs) or the World Health Organization Prequalification of Medicines Programme, programs would be able to procure capreomycin and amikacin, for example, from additional manufacturers (Vianex and Cipla, respectively), even if only during domestic shortages. In addition, programs would be able to procure drugs through the Global Drug Facility (GDF), which is housed in the Stop TB Partnership and mandated to ensure uninterrupted access by national TB programs to high-quality TB drugs. Buying into the GDF would also allow for consolidation of demand—which is key to creating a more robust and predictable market that appeals to drug suppliers—and significant savings, as GDF drugs are much cheaper than those currently procured in the United States.
Barring FDA acceptance of WHO prequalified drugs, the United States could create a parallel, centralized TB drug system that would allow for pooled procurement and more efficient ordering than the current fragmented system in which each program procures independently. It would also monitor supply to prevent shortages or detect them early, and to coordinate distribution in times of shortage. A centralized TB drug procurement and distribution system could be modeled after Texas’s drug procurement program or the CDC’s Vaccines for Children Program. If modeled after the latter, the CDC would consolidate demand, purchase TB drugs, and distribute them to state programs based on need. This method would eliminate inefficiencies that result from the existing fragmented market and allow drug surplus to be reallocated or added to the emergency stockpile. A centralized model that requires continuous communication between state programs and the CDC would help to keep all parties aware of imminent shortages and informed of solutions put in place to minimize the effects on patients.However, establishing a new system would be quite resource-intensive—an important consideration given the current fiscal crisis.
TB drug shortages are a public health emergency that should not be left to market forces for resolution. “I understand there are a lot of challenges for the Obama administration to tackle right now,” said Thoman. “I know there are bigger fish to fry, but I would like to see another presidential executive order directed at the drug shortage. Either the FDA needs to step up and actually figure out how to solve the drug shortage or another entity should be put in charge.”
We can take several immediate steps in the interim:
- Demand needs to be consolidated across states or regions to improve efficiency;
- The FDA needs to enforce legal requirements for drug suppliers to report shortages and impending shortages within a specified time;
- Interagency coordination and communication must be improved;
- The CDC should establish a central emergency stockpile of TB drugs at the U.S. Department of Health and Human Services Supply Center that programs can draw from during shortages; and
- The FDA needs to take accountability and address pricing issues for TB and other drugs, and work with the Department of Justice to reprimand all manufacturers that increase prices during current and future drug shortages.
