Updated April 2016
The goal of hepatitis C virus (HCV) treatment is a cure (when there is no hepatitis C virus in a person’s bloodstream at least 12 weeks after treatment is finished).
What is Harvoni?
Harvoni is two HCV-fighting drugs (sofosbuvir and ledipasvir) in one pill. In the United States, Harvoni is approved for HIV-negative and HIV-positive people with hepatitis C genotypes 1, 4, 5, and 6 who are over 18 years old. Harvoni is also approved for people with HCV genotype 1 who have advanced (called decompensated) cirrhosis, and for liver transplant recipients who have HCV genotype 1 or 4.
How is Harvoni used?
Harvoni is taken once daily, with or without food, for 8 to 24 weeks. Treatment length depends on HCV treatment history, whether a person has cirrhosis, and the amount of hepatitis C virus in a person’s bloodstream (called HCV RNA or viral load). Some people will need to add another drug, called ribavirin (RBV), twice daily with Harvoni.
FDA Recommended Treatment Length and Cure Rates in Clinical Trials*
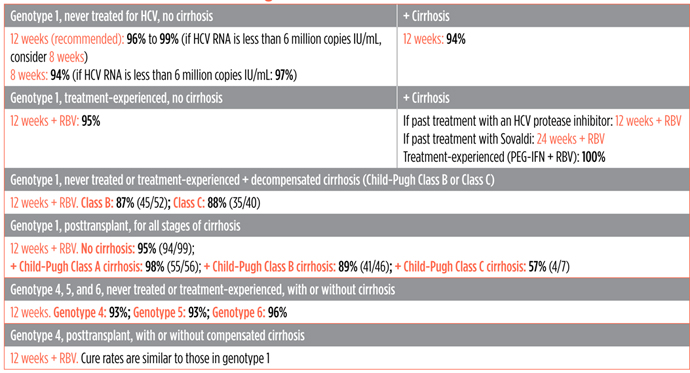
*Cure rates in clinical trials are higher than in real life since the people in them are usually healthier and get extra monitoring and support.
The most important thing a person can do to be cured is not to miss taking doses of HCV treatment—this is called adherence. Adherence lowers the risk for drug resistance.
What is drug resistance?
Each day, HCV makes billions of copies of itself. Some of these copies are not exactly the same as the original virus. They may have changes (called mutations) that can stop hepatitis C drugs from working. If people miss doses of their treatment, HCV gets a chance to reproduce—and some of the copies it makes may not respond to HCV treatment.
Some people have drug resistance even though they have never been on hepatitis C treatment—but many can be cured anyway.
Most people who are not cured have resistance to one or more of the HCV drugs they’ve taken. Resistance to certain hepatitis C drugs can disappear within months. But resistance to certain drugs, including Harvoni, can last for years and may limit re-treatment options.
Harvoni and age, gender, and race/ethnicity:
In clinical trials, cure rates did not differ by age (over 65 vs. under 65). Cure rates have been the same for women and men. There is not much information about how well Harvoni works by race or ethnicity because most of the people in the trials were white. With HCV alone, black (99%, or 89/90) versus nonblack (96%, or 431/448) people were just as likely to be cured by 12 weeks of Harvoni. In ION-4, a trial in HIV/HCV coinfection, the overall cure rate was higher (96%, or 321/335) than among black participants (90%, or 105/115).
Side effects from Harvoni:
Talk with your health care provider about possible side effects and how they will be managed. In clinical trials of Harvoni, the most common side effects were fatigue, headache, nausea, diarrhea, and insomnia; usually, these were mild. Some people have reported skin swelling, rash, or blisters.
Does Harvoni work for HIV-positive people?
Yes. In clinical trial of 335 HIV/HCV-coinfected people, 321 (96%) were cured after 12 weeks of Harvoni. Harvoni cannot be used with certain HIV drugs (see Harvoni and other medications, below).
Harvoni and other medications:
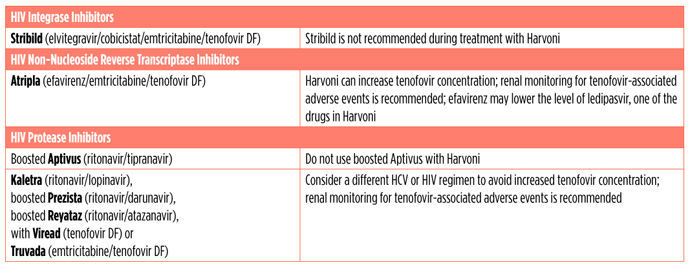
Harvoni should not be used with certain drugs. Combining medications can increase or lower drug levels (called drug-drug interactions). Increasing drug levels can make side effects from each drug worse. If drug levels get too low, a drug can stop working, putting people at risk for drug resistance or not being cured.
Harvoni should not be used with a medication called amiodarone because it can cause life-threatening heart problems. For people who must take amiodarone, intensive heart monitoring in a hospital is recommended for 48 hours after starting Harvoni, and daily monitoring for at least 2 weeks afterwards.
Talk with your health care provider before starting or stopping any medications, supplements, or herbal remedies.
Some drugs should be switched, stopped or avoided while using Harvoni. More information is available in Harvoni’s prescribing information (http://www.accessdata.fda.gov/drugsatfda_docs/label/2014/ 205834s000lbl.pdf) and at: www.hep-druginteractions.org.
Harvoni and HIV Antiretrovirals
Storing Harvoni:
Keep Harvoni at room temperature (below 86°F).
Harvoni in people with kidney disease:
Harvoni can be used in people with mild or moderate kidney disease. It is not recommended for people with severe kidney disease (eGFR<30 mL/min/1.73 m2) or for people on dialysis.
Harvoni in people with cirrhosis:
HCV treatment guidelines recommend that people with serious liver damage (Child-Pugh Class B or C) be treated by a specialist. Harvoni has been approved for use in compensated (Child-Pugh Class A) and decompensated (Child-Pugh Class B or Class C cirrhosis).
Harvoni during pregnancy, nursing, and in children:
It is not known whether Harvoni causes harm to unborn babies. If you are pregnant or planning pregnancy, talk with your health care provider about the risks and benefits of HCV treatment. It is not known whether Harvoni passes into breast milk. Harvoni is under study in children (ages 3 to 18).
Ribavirin causes birth defects, and it can be fatal to unborn babies. Ribavirin should not be used by pregnant women, or by male partners of pregnant women. Ribavirin stays in a person’s body for months. Women and their male partners should avoid pregnancy for six months after they have stopped taking ribavirin. Using two forms of birth control to prevent pregnancy while taking ribavirin—and for six months afterwards—is recommended. Nursing during treatment with ribavirin is not recommended. (See the Ribavirin Fact Sheet for more information)
There is a ribavirin pregnancy registry at: http://www.ribavirinpregnancyregistry.com.
Access to Harvoni
Access may be restricted by public and private payers. The criteria differ depending on the type of coverage and the state it is issued in. Support Path is Gilead’s patient assistance program for Harvoni. People with private insurance may be eligible for assistance with copayments. Uninsured people may be eligible for medication at no charge. Information about Support Path is available online at: http://www.gilead.com/responsibility/us-patientaccess/ support%20path%20for%20sovaldi%20and%20harvoni. Information about Support Path is also available by phone at 1.855.769.7284, Monday through Friday between 9:00 a.m. and 8:00 p.m. (Eastern Time), or online at: https://www.harvoni.com/support.
This fact sheet is current as of April 2016. Always check for updated information.

