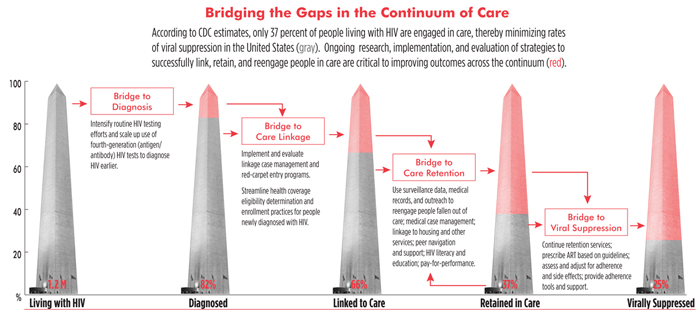
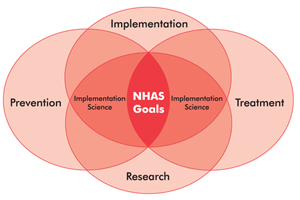
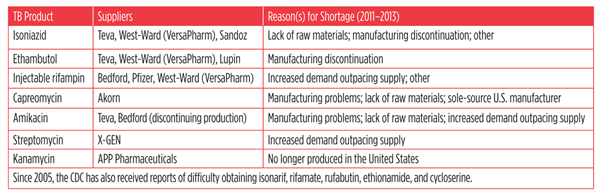
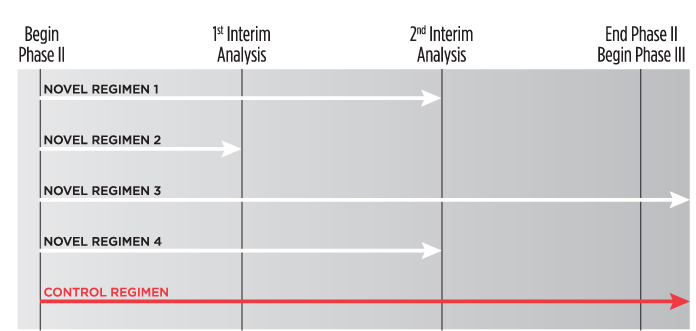
Toward a Plan to End AIDS in New York State
A coalition of community groups push to end AIDS at the epicenter of the U.S. epidemic Beginning in January 2013, a coalition of New York HIV/AIDS leaders came together to begin a series of discussions to reenvision the state’s HIV/AIDS…